

You can:
| Name | Muscarinic acetylcholine receptor M5 |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | CHRM5 |
| Synonym | M5R M5 receptor cholinergic receptor cholinergic receptor, muscarinic 5 |
| Disease | Urinary incontinence Colitis Dysmenorrhea Irritable bowel syndrome Myasthenia gravis [ Show all ] |
| Length | 532 |
| Amino acid sequence | MEGDSYHNATTVNGTPVNHQPLERHRLWEVITIAAVTAVVSLITIVGNVLVMISFKVNSQLKTVNNYYLLSLACADLIIGIFSMNLYTTYILMGRWALGSLACDLWLALDYVASNASVMNLLVISFDRYFSITRPLTYRAKRTPKRAGIMIGLAWLISFILWAPAILCWQYLVGKRTVPLDECQIQFLSEPTITFGTAIAAFYIPVSVMTILYCRIYRETEKRTKDLADLQGSDSVTKAEKRKPAHRALFRSCLRCPRPTLAQRERNQASWSSSRRSTSTTGKPSQATGPSANWAKAEQLTTCSSYPSSEDEDKPATDPVLQVVYKSQGKESPGEEFSAEETEETFVKAETEKSDYDTPNYLLSPAAAHRPKSQKCVAYKFRLVVKADGNQETNNGCHKVKIMPCPFPVAKEPSTKGLNPNPSHQMTKRKRVVLVKERKAAQTLSAILLAFIITWTPYNIMVLVSTFCDKCVPVTLWHLGYWLCYVNSTVNPICYALCNRTFRKTFKMLLLCRWKKKKVEEKLYWQGNSKLP |
| UniProt | P08912 |
| Protein Data Bank | N/A |
| GPCR-HGmod model | N/A |
| 3D structure model | No available structures or models |
| BioLiP | N/A |
| Therapeutic Target Database | T79961 |
| ChEMBL | CHEMBL2035 |
| IUPHAR | 17 |
| DrugBank | BE0000247, BE0004890 |
| Name | carbachol |
|---|---|
| Molecular formula | C6H15ClN2O2 |
| IUPAC name | 2-carbamoyloxyethyl(trimethyl)azanium;chloride |
| Molecular weight | 182.648 |
| Hydrogen bond acceptor | 3 |
| Hydrogen bond donor | 1 |
| XlogP | None |
| Synonyms | HMS2236L04 Carbachol,(S) Isopto Carbachol Carbacholum chloratum Lentine [ Show all ] |
| Inchi Key | AIXAANGOTKPUOY-UHFFFAOYSA-N |
| Inchi ID | InChI=1S/C6H14N2O2.ClH/c1-8(2,3)4-5-10-6(7)9;/h4-5H2,1-3H3,(H-,7,9);1H |
| PubChem CID | 5831 |
| ChEMBL | CHEMBL14 |
| IUPHAR | N/A |
| BindingDB | N/A |
| DrugBank | DB00411 |
Structure | 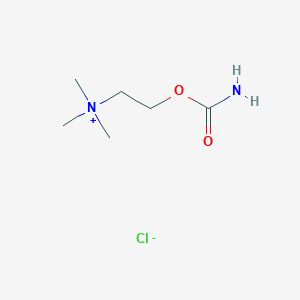 |
| Lipinski's druglikeness | Partition coefficient log P of this ligand is not available. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Activity | 14.7 /s | PMID20716489 | ChEMBL |
| Activity | 200.0 % | PMID13678406 | ChEMBL |
| EC50 | 360.0 nM | PMID7783150 | ChEMBL |
| EC50 | 1000.0 nM | PMID17149881 | ChEMBL |
| EC50 | 2800.0 nM | PMID20716489 | ChEMBL |
| EC50 | 6309.57 nM | PMID13678406 | ChEMBL |
| EC50 | 8900.0 nM | PMID11741475 | ChEMBL |
| EC50 | 9.12011e+13 nM | PMID17084634 | ChEMBL |
| ED50 | 2.7 uM | PMID9651157 | ChEMBL |
| ED50 | 1400.0 nM | PMID9651157 | ChEMBL |
| IC50 | 25200.0 nM | PMID9873472 | ChEMBL |
| Intrinsic activity | 1.0 - | PMID17084634 | ChEMBL |
| Ki | 4.3 nM | PMID12747793 | PDSP |
| Ki | 4265.8 nM | PMID18182302 | ChEMBL |
| Ki | 4300.0 nM | PMID12747793 | ChEMBL |
| Ki | 19952.6 nM | PMID17149881, PMID13678406 | ChEMBL |
| Ki | 69183.1 nM | PMID18543900, PMID24980056, PMID19896386, PMID16539379, PMID18077164 | ChEMBL |
| PI | 100.0 % | Bioorg. Med. Chem. Lett., (1995) 5:6:631 | ChEMBL |
| Smax | 110.0 % | PMID11741475 | ChEMBL |
| Stimulation | 100.0 % | PMID9651157 | ChEMBL |
| TIME | 1.889e-05 hr | PMID20716489 | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218